Pancreatic cancer (also known as PaC) begins in the tissues of the pancreas, an organ in the abdomen that lies behind the lower part of the stomach. The pancreas releases enzymes that aid digestion and produces hormones that help control blood glucose.
Several types of tumors can occur in the pancreas, including cancerous and noncancerous tumors. In 95% of cases, the most common type of cancer that forms in the pancreas begins in the cells that line the ducts that carry digestive enzymes out of the pancreas (pancreatic ductal adenocarcinoma or PDAC).
The most important and well-known precursor of a pancreatic ductal adenocarcinoma (PDAC) is pancreatic intraepithelial neoplasia (PanIN), a microscopic (usually <5 mm) flat or papillary lesion arising in the small intralobular pancreatic ducts.
Some growths in the pancreas are simply benign (not cancer), while others might become cancer over time if left untreated (known as precancers). Since people are getting imaging tests such as Computed Tomography scans more often than in the past (for a number of reasons), these types of pancreatic growths are now being found more often. The following cystic lesions in the pancreas can become cancer in the future:
Mucinous cystic neoplasms (MCNs) (also known as mucinous cystadenomas) are slow-growing tumors that have cysts filled with a jelly-like substance called mucin. While they are not cancer, some of them can progress to cancer over time if not treated, so these tumors are typically removed with surgery
Intraductal papillary mucinous neoplasms (IPMNs) are benign tumors that grow in the pancreatic ducts. Like MCNs, these tumors make mucin, and over time they sometimes become cancer if not treated.
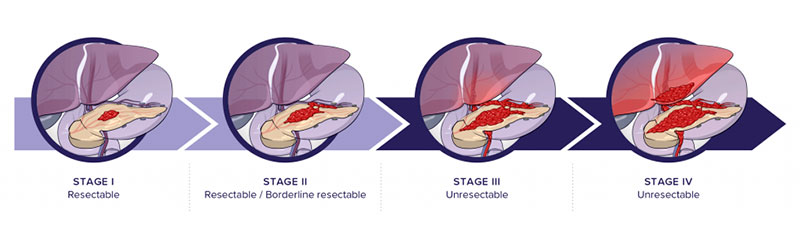
Pancreatic cancer is one of the more aggressive neoplasms and has a worse prognosis due to its early dissemination, lack of specific symptoms in early stages and, therefore, a late diagnosis. Generally, at the time of diagnosis, patients present locally advanced disease and in many cases distant metastasis, which prevents curative surgery. Early detection, understood as a diagnosis of the disease in operable stages, is therefore essential to improve patient survival.
Incidence
It is estimated that in 2025, 557,688 new cases of pancreatic cancer will be diagnosed worldwide. Unlike what happens in other types of tumours where screening procedures significantly reduce late diagnosis, in pancreatic cancer there are currently no resolute imaging techniques or molecular methods that allow the disease to be detected before patients present symptoms.
Mortality
Pancreatic cancer, despite being the 7th most frequent tumor in industrialized countries (constituting 2% to 3% of all solid tumors), represents the 4th cause of death from cancer and by 2030 its incidence is expected to increase by 50%, becoming the 2nd leading cause death from cancer and causing more deaths than prostate, colon, or breast cancer.
Because of the lack of early symptoms, aggressive growth and early dissemination, 80% of patients are diagnosed at late stages with advanced distant metastases, which make their disease surgically inoperable. Survival at stage IV is less than 5%.
Surgery is the only treatment considered curative, but only 20% of diagnosed patients have this option. Unfortunately, most patients have locally advanced or metastatic disease at diagnosis and are therefore not candidates for surgery.
The low survival rate is related to several factors, the late stage at which patients are diagnosed probably being the most important of all. It is essential to be able to detect PaC early to reduce the high mortality associated with this disease.

Diagnosis
As mentioned above, pancreatic cancer is a silent disease in the initial phase and becomes evident when the tumour infiltrates neighbouring and distant organs. The typical symptoms are abdominal and lower back pain, jaundice, and weight loss.
Symptomatic patients are diagnosed by non-invasive imaging techniques, which often reveal a pancreatic mass, in conjunction with invasive techniques to obtain a biopsy specimen for diagnostic intent. These techniques reliably detect 2 cm tumours, although in some cases they can reach 1 cm.
- Transabdominal ultrasound is usually the technique applied for the initial examination, with good sensitivity for tumours larger than 3 cm.
- Computed tomography (CT) is the best initial diagnostic test and also for local and remote staging, its sensitivity varying depending on the location of the tumour.
- Endoscopic ultrasound (EUS) is a technique that serves both for diagnosis and for sampling, necessary for an accurate diagnosis.
- Endoscopic ultrasound-guided fine needle aspiration-puncture (EUS-FNA) is the most widely used modality for obtaining samples with a sensitivity greater than 80%. It can also be CT-guided.
- Magnetic resonance imaging provides good information on the relationship of the tumour with neighbouring structures. It is not used for staging.
Finally, there is also a protein biomarker, the CA19.9 antigen, that is detected by biochemical methods, although it has relatively low rates of sensitivity and specificity values (79% and 82%, respectively) for diagnostic use. Currently it is used mainly for follow-up and relapse detection.